Membrane filtration principle
Membrane filtration technology is based on a selective filtration through a pressure-driven membrane for sulfate water treatment. The membrane has charged groups that selectively reject multivalent anions such as SO42-. The membrane does not separate monovalent ionic species such as Cl- or CLO3- from the feed solution.
To maximize the membrane area within a compact space, the membranes are packaged as cylindrical elements in a spiral wound configuration.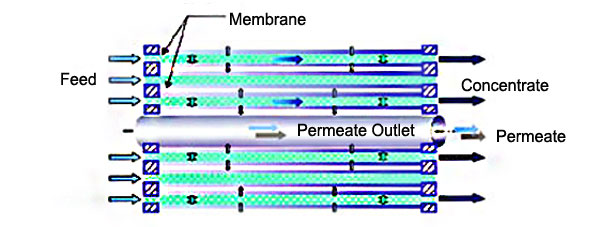
Feed brine floods the entire feed side of the membrane surface within the membrane element by flowing within a mesh spacer. Brine then permeates through the membrane, where the sulfate is rejected, and flows in a spiral pattern through the permeate carrier, towards the perforated perforated permeate tube.
To achieve the required membrane area, several membrane elements are packed into several high-pressure housing. The high-pressure housings are arranged in series to provide the required number of stages to concentrate the sodium Sulfate solution.
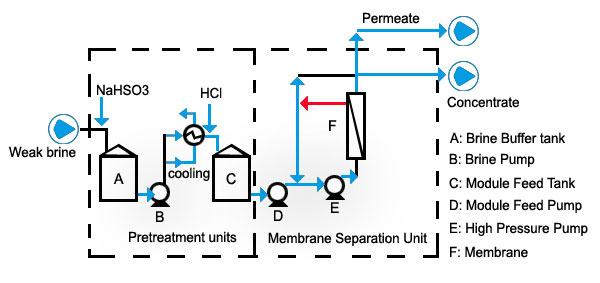
The ceramic membrane filtration unit usually treats dechlorinated depleted brine from the chlor-alkali electrolyzer. It is at this location that athe Na2SO4 concentration is at its highest and the NaCL concentration is at its lowest. Normally only a portion of the dechlorinated brine flow needs to be passed through Sulfate removal system, the remainder bypasses the Sulfate removal unit and is returned to the saturators..jpg)
.jpg)
Features:
1. Reduced purge of depleted brine
Substantial costs saving due to less NaCL loss
More Environmental friendly, less effluent
2. No secondary pollution
No need to use BaCl2 reagent which is toxic
No Barium treatment cost
No solid BaSO4 waste
3. No secondary pollution
Lower operating costs
Reduces effluent volume
Full recovery of sulfate and sodium chloride
Proven technology with many references



we will contact you within 24 hours.
 +86-25-58849045
+86-25-58849045
 +86-25-58749295
+86-25-58749295
 jiuwu@jiuwu.com
jiuwu@jiuwu.com
 No. 9 Park Road, Pukou District, Nanjing City (Sanqiao Factory)
No. 9 Park Road, Pukou District, Nanjing City (Sanqiao Factory) Call us on:
Call us on:  Email Us:
Email Us:  No. 9 Park Road, Pukou District, Nanjing City (Sanqiao Factory)
No. 9 Park Road, Pukou District, Nanjing City (Sanqiao Factory)

 English
English 한국어
한국어 français
français русский
русский Español
Español



